The short answer is yes. Recent research indicates that SCFAs—particularly butyrate—play a significant, multi-faceted role in protecting pancreatic function and health.
The benefits are not just general “gut health” spillover; there are direct molecular mechanisms at play in pancreatic cells.
1. The Core Mechanism: HDAC Inhibition
To understand how SCFAs help the pancreas, you need to look at Histone Deacetylases (HDACs).
- The Problem: In conditions like pancreatitis or Type 2 Diabetes, inflammatory cytokines (like IL-1β) attack pancreatic cells. They effectively “lock down” the genes responsible for insulin production and repair.
- The Solution: Butyrate acts as a natural HDAC inhibitor. By inhibiting these enzymes, butyrate prevents the DNA in pancreatic cells from being “locked up.” This keeps the chromatin “open,” allowing the cells to continue reading the genes necessary for:
- Beta-cell differentiation (keeping insulin-producing cells mature and functional).
- Insulin secretion (maintaining the ability to respond to glucose).
- Anti-inflammatory defense (blocking the NF-κB pathway that drives tissue damage).
2. Benefits for Specific Conditions
A. Acute & Chronic Pancreatitis
Pancreatitis is often made worse when the gut barrier fails, allowing bacteria to leak into the bloodstream (translocation), causing sepsis.
- Gut Barrier Shield: SCFAs strengthen the “tight junctions” of the gut lining, preventing this bacterial leakage.
- Direct Anti-Inflammatory: Studies using Sodium Butyrate (a supplemental form) have shown it can reduce the severity of acute pancreatitis by lowering systemic inflammatory markers like TNF-α and IL-6. It effectively dampens the “cytokine storm” that damages the pancreas.
B. Type 2 Diabetes & Insulin Resistance
SCFAs appear to be a missing link in why fiber prevents diabetes.
- Beta-Cell Bodyguard: Research suggests butyrate protects pancreatic beta-cells from apoptosis (cell death) induced by metabolic stress.
- Insulin Secretion: Propionate and butyrate stimulate the release of GLP-1 (a hormone that boosts insulin secretion) from the gut. This is the same pathway targeted by drugs like Ozempic, but SCFAs trigger it naturally via dietary fermentation.
C. Pancreatic Cancer (Emerging Research)
While this is a complex field, dysbiosis (lack of healthy bacteria) is a known risk factor for Pancreatic Ductal Adenocarcinoma (PDAC).
- Antiproliferative Effect: In lab settings, SCFAs have been shown to stop the proliferation of pancreatic cancer cells and induce apoptosis (programmed cell death), again likely via the HDAC inhibition pathway.
- Tumor Microenvironment: They may help recruit cancer-fighting immune cells (CD8+ T cells) into the tumor environment, making the immune system more effective at recognizing and attacking abnormal cells.
3. Practical Application: Diet vs. Supplements
Given your background with VitaNet, it is worth distinguishing between the two primary delivery methods found in the research:
| Method | Source | Benefit Profile |
| Endogenous Production | Prebiotic Fibers: Inulin, Resistant Starch, Pectin. | Sustained Release: Produces a steady stream of SCFAs (Acetate, Propionate, Butyrate) along the entire colon. Best for preventative health and metabolic signaling. |
| Exogenous Supplementation | Sodium Butyrate: Encapsulated salts (often enteric-coated). | Targeted Therapy: Delivers a higher, more potent dose directly to the lower gut. This is the form often used in studies showing rapid recovery from pancreatitis or acute inflammation. |
Summary for Your Business Context
Gut health is pancreatic health! Feeding the gut microbiome (or supplementing butyrate directly), provide the pancreas with the molecular “brakes” (HDAC inhibitors) it needs to stop inflammation and the “fuel” to maintain insulin production.
The Gut-Pancreas Axis” and the role of Butyrate?
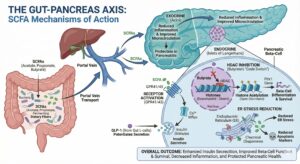
Lets look at the specific signaling pathways and molecular targets that Short-Chain Fatty Acids (SCFAs)—specifically Butyrate, Propionate, and Acetate—engage within the pancreas.
This interaction is commonly referred to in literature as the Gut-Pancreas Axis. It operates through three distinct mechanisms: direct receptor activation, epigenetic modification (HDAC inhibition), and systemic metabolic regulation.
1. The Delivery System: The Portal Vein Pathway
Before understanding the mechanism, it is crucial to understand the route. When fibers are fermented in the colon, the resulting SCFAs are not just used locally by colonocytes.
- Uptake: They are absorbed into the bloodstream and travel via the hepatic portal vein.
- Significance: This vein goes directly to the liver and the pancreas before reaching the rest of the systemic circulation. This means the pancreas is exposed to significantly higher concentrations of SCFAs than peripheral tissues (like muscles), making it a primary target for SCFA therapy.
2. Mechanism A: G-Protein Coupled Receptors ( The “Doorbell” Effect)
Pancreatic cells possess specific sensors on their surface designed to detect SCFAs. These are Free Fatty Acid Receptors (FFARs), specifically GPR41 (FFAR3) and GPR43 (FFAR2).
When SCFAs bind to these receptors on pancreatic beta-cells, they trigger a cascade:
- Intracellular Calcium Influx: The binding causes an increase in cytosolic Ca2+.
- Insulin Granule Release: Insulin is stored in granules inside the cell. The calcium spike signals these granules to fuse with the cell membrane and release insulin into the blood.
- The “Incretin” Boost: SCFAs also stimulate L-cells in the gut to release GLP-1 (Glucagon-like peptide-1). GLP-1 travels to the pancreas and further potentiates insulin secretion.
The Clinical Implication: This suggests that SCFAs act as a “glucose-dependent” insulin secretagogue—meaning they help the pancreas release insulin when it is needed, reducing the risk of hypoglycemia (low blood sugar) compared to harsh pharmaceutical drugs.
3. Mechanism B: HDAC Inhibition & Gene Expression (The “Code Switch”)
This is perhaps the most profound mechanism, particularly for Butyrate.
Inside the nucleus of a pancreatic cell, DNA acts as the instruction manual for building proteins (like insulin or repair enzymes). This DNA is wrapped around spools called histones.
- Deacetylated Histones (Closed): When histones are “deacetylated,” the DNA is wrapped too tightly. The cell cannot read the instructions. This happens during chronic inflammation and stress.
- Acetylated Histones (Open): Butyrate inhibits the enzyme (HDAC) that tightens the spool. This keeps the DNA “open” (acetylated).
What genes get turned on?
- Pdx1: This is the “master switch” for pancreatic development. Keeping this gene active is vital for maintaining beta-cell identity (preventing them from forgetting they are beta-cells).
- Anti-apoptotic genes: Genes that tell the cell not to self-destruct under stress.
4. Mechanism C: Reducing Endoplasmic Reticulum (ER) Stress
A major cause of pancreatic failure in Type 2 Diabetes is ER Stress. The beta-cells are forced to churn out so much insulin that their protein-folding machinery (the Endoplasmic Reticulum) gets overwhelmed. Misfolded proteins accumulate, becoming toxic (amyloid deposits), eventually killing the cell.
How SCFAs help:
- Research indicates that Butyrate and Phenylbutyrate act as Chemical Chaperones. They stabilize protein folding within the ER.
- They reduce the expression of pro-apoptotic markers (like CHOP) that are usually triggered when the ER is stressed.
Basically, SCFAs help the pancreas handle a heavy workload without burning out.
5. Impact on Exocrine Health (Pancreatitis)
The pancreas also produces digestive enzymes (exocrine function). In Acute Pancreatitis, these enzymes activate inside the pancreas, effectively causing the organ to digest itself.
- Microcirculatory Support: Severe pancreatitis causes blood flow constriction (ischemia) to the organ, leading to necrosis (tissue death). SCFAs have been shown to improve pancreatic microcirculation, keeping blood flowing to the damaged tissue to aid repair.
- Neutrophil Migration: SCFAs modulate the immune system to prevent an excessive influx of neutrophils (white blood cells) into the pancreas. While you need some immune response to heal, too many neutrophils release enzymes that liquefy pancreatic tissue. SCFAs keep this response balanced.
Summary of Actionable Biological Targets
| SCFA Component | Primary Target | Physiological Outcome |
| Butyrate | HDAC Enzymes | Prevents beta-cell death; reduces inflammation; maintains cell identity. |
| Propionate | GPR43 Receptor | Stimulates GLP-1 release; improves insulin sensitivity. |
| Acetate | Parasympathetic NS | Activates the vagus nerve to promote insulin secretion via the gut-brain-pancreas axis. |
In Summary:
The Gut-Pancreas Axis and how Short-Chain Fatty Acids (SCFAs) support pancreatic function:
The Core Concept
The Gut-Pancreas Axis describes the biological link where gut bacteria ferment dietary fiber into SCFAs (Butyrate, Propionate, Acetate). These molecules are not just fuel for the gut; they travel via the hepatic portal vein directly to the pancreas to regulate its function and protect it from damage.
Three Key Mechanisms of Action
- The “Code Switch” (HDAC Inhibition):
- Agent: Primarily Butyrate.
- Action: It inhibits Histone Deacetylase (HDAC) enzymes. This prevents pancreatic DNA from being “locked up,” keeping essential genes (like Pdx1) open and active.
- Result: Maintains beta-cell identity (so they don’t forget how to make insulin) and prevents cell death (apoptosis).
- The “Doorbell” (Receptor Activation):
- Agent: Propionate and Acetate.
- Action: They bind to specific receptors (GPR41/43) on the surface of pancreatic cells.
- Result: This triggers a calcium influx that signals the release of insulin. It also stimulates the gut to release GLP-1, further boosting insulin sensitivity (similar to how modern diabetes drugs work, but naturally).
- The “Stress Relief” (ER Stress Reduction):
- Action: SCFAs act as “chemical chaperones” inside the cell.
- Result: They help the Endoplasmic Reticulum (ER) fold proteins correctly, preventing the beta-cell “burnout” common in Type 2 Diabetes.
Health Outcomes
- For Diabetes: Improved insulin secretion and protection of beta-cells from dying off.
- For Pancreatitis: Reduced inflammation (lower cytokines) and improved blood flow (microcirculation) to damaged tissue, preventing necrosis.
Practical Takeaway
To activate this axis, one must either consume prebiotic fibers (to feed the bacteria that make SCFAs) or supplement with exogenous SCFAs (like Sodium Butyrate) to deliver the therapeutic molecules directly to the portal vein.
Leave a Reply