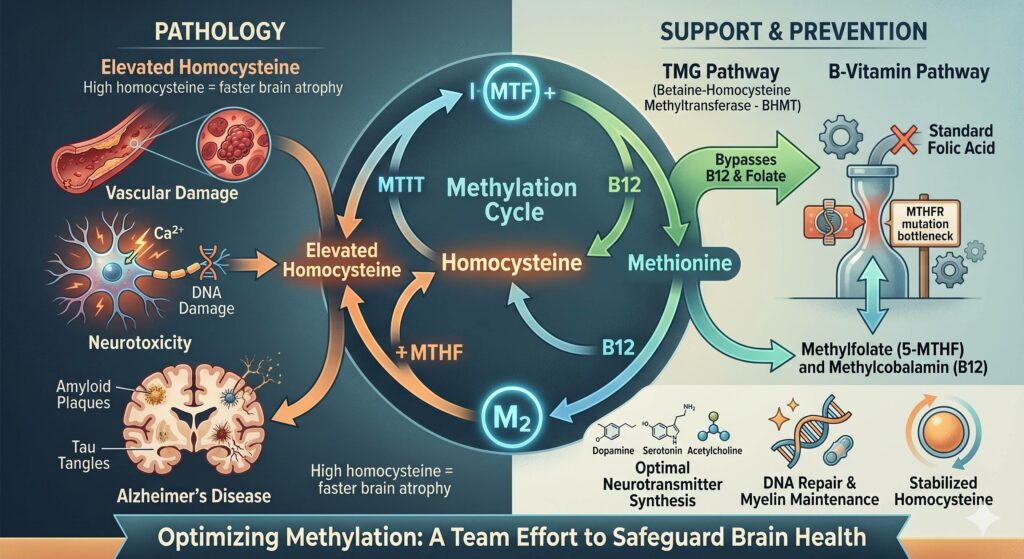
The relationship between methylation and homocysteine levels is a critical factor in neurology. When the methylation cycle is impaired, homocysteine levels rise, creating a biochemical environment that can contribute to cognitive decline and the pathology of Alzheimer’s disease.
1. The Methylation-Homocysteine Connection
Methylation is a fundamental biochemical process where a “methyl group” (CH3) is transferred to various molecules to regulate gene expression, neurotransmitter production, and protein function.
Homocysteine is an amino acid produced as a byproduct of this cycle. In a healthy system, homocysteine is quickly converted back into methionine (via B12 and folate) or into cysteine (via B6). If the methylation cycle “stalls” due to nutrient deficiencies or genetic factors, homocysteine accumulates in the blood, leading to a condition called hyperhomocysteinemia.
2. Impact on Brain Health
High homocysteine levels affect the brain through several destructive mechanisms:
- Neurotoxicity: Elevated homocysteine acts as an excitotoxin, overstimulating NMDA receptors in the brain. This can lead to an influx of calcium into neurons, causing oxidative stress and eventually cell death (apoptosis).
- Vascular Damage: Homocysteine damages the lining of blood vessels (the endothelium). This increases the risk of “silent” micro-strokes and reduced blood flow to the brain, which are primary drivers of vascular dementia.
- DNA Damage and Repair: Methylation is required to repair DNA. Poor methylation status leaves neurons vulnerable to genetic “glitches” and reduces the brain’s ability to recover from standard metabolic stress.
3. Role in Alzheimer’s Disease
Research has consistently linked high homocysteine to the hallmarks of Alzheimer’s disease:
| Feature | Impact of High Homocysteine / Poor Methylation |
| Amyloid Plaques | High homocysteine levels are associated with increased production and decreased clearance of amyloid-beta, the protein that forms plaques in Alzheimer’s. |
| Tau Tangles | Impaired methylation can lead to the “hyperphosphorylation” of tau proteins, which causes the internal structure of neurons to collapse into tangles. |
| Brain Atrophy | Clinical studies, such as the VITACOG trial, have shown that individuals with high homocysteine experience a faster rate of brain shrinkage (atrophy), particularly in the hippocampus. |
4. Neurotransmitter Synthesis
Beyond structural damage, methylation is responsible for the production of key neurotransmitters:
- Dopamine & Serotonin: These rely on methyl donors for their synthesis and metabolism.
- Acetylcholine: Essential for memory and learning; its production requires methyl groups.
- Myelin: The protective coating around nerves (myelin) requires constant methylation for maintenance. If methylation is poor, signal transmission between neurons slows down.
Summary of the “B-Vitamin Hypothesis”
Because Vitamin B12, Folate (B9), and Vitamin B6 are the primary cofactors for the methylation cycle, much of the research on preventing dementia focuses on these nutrients. When these levels are optimal, homocysteine is kept low, which may offer a neuroprotective effect against the biological markers of aging and cognitive decline.
TMG can also boost Methylation Pools
TMG (Trimethylglycine) and specific form of B vitamins play pivotal roles in managing homocysteine, especially when genetic variations are present. Here is a breakdown of how they function as a “backup system” and why the “methylated” label matters.
1. TMG: The Alternative Pathway
While the primary way the body lowers homocysteine is through the folate and B12 pathway, TMG (also known as Betaine) provides a crucial “shortcut.”
- The BHMT Shortcut: In the liver and kidneys, an enzyme called Betaine-Homocysteine Methyltransferase (BHMT) can convert homocysteine back into methionine. Unlike the main pathway, this process does not require B12 or Folate.
- Direct Methyl Donation: TMG has three methyl groups. It “donates” one directly to homocysteine to neutralize it.
- Synergy: TMG is often used alongside B vitamins because it acts as a safety net. If the B-vitamin pathway is sluggish due to stress or genetics, the TMG pathway can help pick up the slack to prevent homocysteine from spiking.
2. Genetic Mutations (MTHFR) and B Vitamins
The concern regarding “methylated” B vitamins usually centers on the MTHFR gene (Methylenetetrahydrofolate Reductase). This gene provides instructions for making an enzyme that converts folate into its final, active form.
Why Methylated Forms are Used
If a person has a mutation in the MTHFR gene (such as the C677T or A1298C variants), the body’s ability to process standard folic acid or food folate is significantly reduced.
- Bypassing the Bottleneck: Standard Folic Acid (synthetic) must go through four enzymatic conversions to become usable. If the MTHFR enzyme is underperforming, the process stalls. Methylfolate (5-MTHF) is the “active” form; taking it bypasses the broken enzyme entirely, delivering the nutrient in a form the body can use immediately.
- B12 (Methylcobalamin): Similarly, Methylcobalamin is a “pre-methylated” form of B12. While the body can convert other forms (like Cyanocobalamin), using the methylated version reduces the metabolic “work” the body has to do, which is particularly helpful if the methylation cycle is already compromised.
3. The “Cofactor” Requirement
Lowering homocysteine is a team effort involving several nutrients. If one is missing, the others may not work effectively:
| Nutrient | Role in the Methylation Cycle |
| Methylfolate (B9) | The primary methyl donor for the “main” recycling pathway. |
| Methylcobalamin (B12) | Acts as the essential cofactor that allows Folate to “hand off” its methyl group to homocysteine. |
| Pyridoxal-5-Phosphate (B6) | Redirects homocysteine down a different path to create Glutathione (the body’s master antioxidant). |
| TMG (Betaine) | Operates the secondary “shortcut” pathway in the liver. |
Why “Methylated” Isn’t Always the Only Factor
While methylated vitamins are highly bioavailable, the effectiveness of the cycle also depends on Riboflavin (B2), which is a required cofactor for the MTHFR enzyme itself, and Magnesium, which is necessary for the production of COMT (an enzyme that uses those methyl groups once they are created).
When these pathways are optimized, homocysteine is efficiently recycled into Methionine and SAMe (S-Adenosylmethionine), which then go on to support brain health, neurotransmitter balance, and cellular repair.
Methylation is a fundamental biochemical process that regulates neurotransmitter production, DNA repair, and the maintenance of the myelin sheath. A critical byproduct of this cycle is homocysteine; when methylation is impaired, homocysteine levels rise, leading to a state of hyperhomocysteinemia. This elevation is neurotoxic, causing oxidative stress and vascular damage that contributes to brain atrophy and the hallmark plaques and tangles of Alzheimer’s disease. Efficient methylation is therefore essential for preserving cognitive function and preventing the structural degradation of the brain.
To counteract high homocysteine, especially in individuals with MTHFR genetic mutations, the body utilizes specific nutrient pathways involving TMG (Betaine) and methylated B vitamins. TMG provides a vital “shortcut” in the liver that recycles homocysteine into methionine independently of the folate pathway. Meanwhile, methylated forms of B12 and folate (5-MTHF) bypass the enzymatic bottlenecks created by genetic variations, ensuring the methylation cycle remains active. Together, these compounds stabilize homocysteine levels, supporting the production of memory-linked neurotransmitters like acetylcholine and protecting overall neurological health.


Leave a Reply