The short answer is yes, but the relationship isn’t always a straight line. It is more like two high-performance engines sharing the same fuel line; when one stutters (under-methylation), the other (NAD+ production) often loses pressure.
The link between under-methylation and low NAD+ levels typically boils down to three metabolic “bottlenecks”:
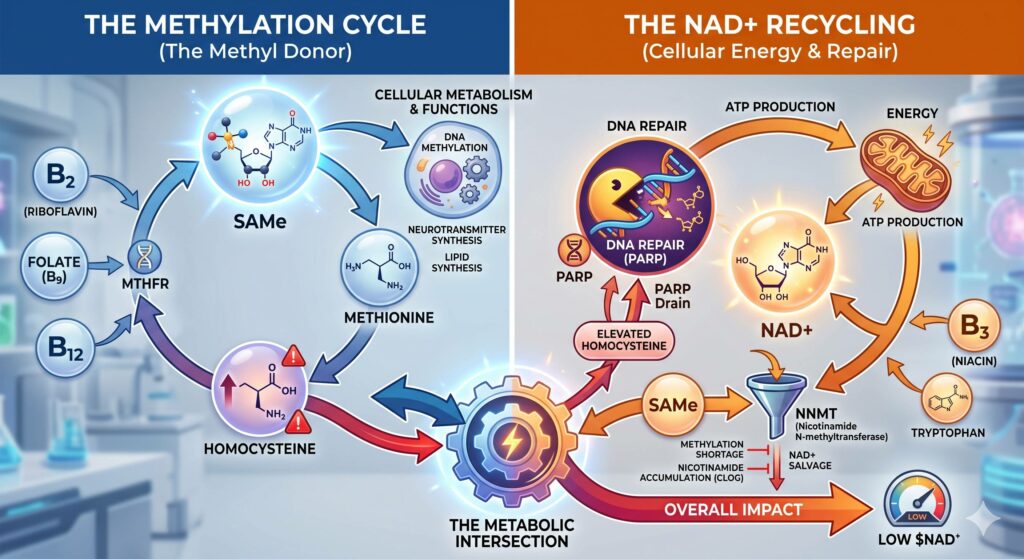
1. The Shared B-Vitamin Pool
Both the methylation cycle and the NAD+ salvage pathway are “B-vitamin hungry.”
- Vitamin B2 (Riboflavin): This is a critical cofactor for MTHFR (the enzyme that drives methylation) and is also required for the conversion of tryptophan into NAD+ and the functioning of the electron transport chain.
- Vitamin B3 (Niacin): This is the direct precursor to NAD+.
- Folate and B12: While these primarily drive methylation, a deficiency here creates a “methyl trap,” leading to metabolic friction that can indirectly drain cellular energy and NAD+ reserves.
If the body is low on these raw materials, you will likely see both sluggish methylation and a dip in NAD+.
2. Oxidative Stress and the “PARP” Drain
This is perhaps the most direct functional link. Under-methylation often leads to an accumulation of homocysteine. High homocysteine is pro-oxidant, meaning it causes “rust” (oxidative stress) within the cells and damages DNA.
When DNA is damaged, the body activates an enzyme called PARP (Poly-ADP Ribose Polymerase) to fix it.
The Catch: PARP is an absolute NAD+ glutton. It consumes massive amounts of NAD+ to perform DNA repair.
If you are under-methylating, you are likely generating more oxidative stress, which keeps PARP “turned on,” effectively hemorrhaging your NAD+ supply to keep up with repairs.
3. The NNMT Connection (The Methylation Sink)
There is an enzyme called NNMT (Nicotinamide N-methyltransferase) that acts as a bridge between these two worlds. Its job is to take excess nicotinamide (a form of B3) and “tag” it with a methyl group from SAMe (the body’s universal methyl donor) so it can be excreted.
- In Under-Methylators: You have a shortage of methyl groups (low SAMe).
- The Result: You may struggle to clear nicotinamide properly. While you might think “more B3 is better,” an accumulation of unprocessed nicotinamide can actually inhibit certain longevity enzymes (like Sirtuins) and disrupt the efficient recycling of NAD+. It creates a “clog” in the salvage pathway rather than a smooth flow.
Summary Table: The Methylation-NAD+ Link
| Factor | Effect of Under-Methylation | Impact on NAD+ |
| Cofactors | Deficit in B2, B3, B6 | Lack of raw materials for NAD+ synthesis. |
| Homocysteine | Levels rise (Hyperhomocysteinemia) | Triggers DNA repair (PARP), which consumes NAD+. |
| SAMe Levels | Low universal methyl donor | Impairs the NNMT pathway and recycling efficiency. |
| ATP Production | Sluggish mitochondrial output | NAD+ synthesis is energy-dependent; low ATP slows it down. |
In essence, if your methylation is “under-performing,” your body is likely redirected away from optimal NAD+ production to deal with the metabolic fallout of high homocysteine and cellular stress. Supporting the methylation cycle with proper methyl donors and B-vitamins often has the side effect of “freeing up” NAD+ for more productive uses, like energy production and cellular aging defense.
In Summary:
Under-methylation and low NAD+ levels are fundamentally linked through shared metabolic precursors and enzymatic bottlenecks. Both processes rely heavily on a specific pool of B-vitamins, particularly Riboflavin (B2) and Niacin (B3), which act as essential cofactors for energy production and methyl transfer. When methylation is sluggish, the body often struggles with a shortage of SAMe, the universal methyl donor. This deficiency disrupts the NNMT pathway, which is responsible for processing nicotinamide; without sufficient methylation, the recycling of NAD+ becomes inefficient, leading to a metabolic “clog” that prevents the salvage pathway from maintaining optimal levels.
Beyond direct nutrient competition, under-methylation creates a significant “drain” on NAD+ through oxidative stress. Poor methylation typically results in elevated homocysteine, which causes cellular damage and DNA fragmentation. This triggers the activation of PARP, a DNA-repair enzyme that is an absolute glutton for NAD+. In an under-methylated state, the body is forced to divert its NAD+ reserves away from mitochondrial energy production and toward constant cellular repair. Consequently, stabilizing the methylation cycle doesn’t just improve methyl donor availability; it also preserves NAD+ by reducing the chronic oxidative demand that otherwise “bleeds” the cellular supply dry.
Leave a Reply