Can Under Methylation Cause Kidney Function Issues? 
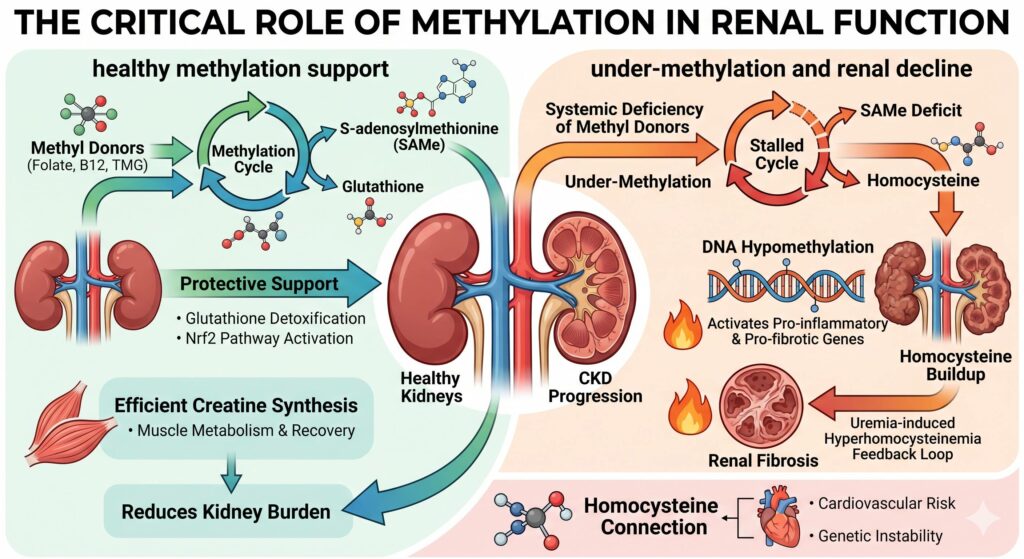
“Under methylation” – clinically referred to as DNA hypomethylation or a systemic deficiency in methyl donors – is significantly linked to the progression of chronic kidney disease (CKD) and the decline of renal function. In the context of the methylation cycle, this often manifests as a lack of available S-adenosylmethionine (SAMe) and an accumulation of homocysteine, both of which can trigger pathological changes in kidney tissue.
1. The Role of Methyl Donor Deficiency
When the body lacks sufficient methyl donors (such as folate, vitamin B12, and TMG), it cannot maintain the “silencing” of certain genes through DNA methylation. In the kidneys, this “under-methylated” state leads to the activation of pro-inflammatory and pro-fibrotic genes (Cappuccilli et al., 2020).
- Renal Fibrosis: Hypomethylation at specific gene promoters can “turn on” pathways that promote the formation of scar tissue (fibrosis) in the kidneys, which is a primary driver of CKD (Cheng, 2025).
- Genetic Instability: Low folate intake specifically results in global DNA hypomethylation, which can disrupt gene expression and undermine the genomic integrity of renal cells (Elworthy, n.d.).
2. The Homocysteine Connection
The relationship between methylation and the kidneys is a “two-way street.” The kidneys are a major site for homocysteine metabolism; when kidney function declines, homocysteine levels typically rise.
- Uremic Impact: Advanced kidney disease leads to “uremia-induced hyperhomocysteinemia,” which inhibits DNA methyltransferases (the enzymes that add methyl groups). This creates a cycle where poor kidney function causes further “under-methylation” throughout the body (Cappuccilli et al., 2020).
- Cardiovascular Risk: High homocysteine in renal patients is a primary contributor to their elevated risk of cardiovascular events (Ingrosso & Perna, 2020).
3. Protective Effects of Methylation Support
Maintaining a robust methylation cycle can protect kidney health by mitigating oxidative stress and supporting detoxification.
- Antioxidant Defense: Adequate methyl donors like 5-MTHF (active folate) have been shown to improve kidney function in models of acute injury. This works by restoring glutathione (the body’s master antioxidant) and activating the Nrf2 pathway, which protects cells from oxidative damage (Wijerathne et al., 2022).
- Recovery and Muscle Metabolism: For those focusing on muscle recovery, the methylation cycle is critical for producing creatine. Since the kidneys share the metabolic burden of creatine synthesis with the liver, optimized methylation reduces the physiological “stress” on these organs during recovery (Wing et al., 2014).
Summary Table: Methylation and Renal Health
| Condition | Effect on Kidney Function |
| DNA Hypomethylation | Activates pro-fibrotic genes, leading to kidney scarring (fibrosis). |
| High Homocysteine | Increases oxidative stress and contributes to vascular damage in the kidneys. |
| Methyl Donor Support | Boosts glutathione levels and activates Nrf2, protecting against injury. |
| Uremia | Interferes with methylation enzymes, worsening systemic “under-methylation.” |
Under-methylation, clinically recognized as DNA hypomethylation, acts as a metabolic trigger for renal decline by “turning on” genes that promote inflammation and fibrosis (scarring) within kidney tissue. When the methylation cycle is compromised, the resulting shortage of methyl donors allows for the accumulation of homocysteine, a toxic byproduct that induces oxidative stress and vascular damage. This creates a destructive feedback loop: as kidney function falters, the body’s ability to clear homocysteine diminishes even further, which in turn inhibits the very enzymes required to maintain healthy DNA methylation, accelerating the progression of chronic kidney disease.
Supporting the methylation cycle with key co-factors helps shield the kidneys by restoring the production of glutathione, the body’s primary antioxidant, and activating the Nrf2 pathway to combat cellular injury. Maintaining adequate methyl donor levels also reduces the physiological burden on the kidneys during creatine synthesis, a resource-intensive process that is vital for metabolic energy and physical recovery. By stabilizing these biochemical pathways, the body can mitigate the genomic instability that leads to renal scarring, ultimately promoting long-term organ health and more efficient systemic detoxification.
References
- Cappuccilli, M., Bergamini, C., Giacomelli, F. A., Cianciolo, G., Donati, G., Conte, D., Natali, T., La Manna, G., & Capelli, I. (2020). Vitamin B Supplementation and Nutritional Intake of Methyl Donors in Patients with Chronic Kidney Disease: A Critical Review of the Impact on Epigenetic Machinery. Nutrients, 12(5), 1234. https://doi.org/10.3390/nu12051234
- Cited by: 52
- Cheng, Y. (2025). DNA methylation in chronic kidney disease. PMC.
- Cited by: 4
- Ingrosso, D., & Perna, A. F. (2020). DNA Methylation Dysfunction in Chronic Kidney Disease. Genes, 11(7), 811. https://doi.org/10.3390/genes11070811
- Cited by: 32
- Wijerathne, C. U. B., Au-Yeung, K. K. W., Siow, Y. L., & O, K. (2022). 5-Methyltetrahydrofolate Attenuates Oxidative Stress and Improves Kidney Function in Acute Kidney Injury through Activation of Nrf2 and Antioxidant Defense. Antioxidants, 11(6), 1046. https://doi.org/10.3390/antiox11061046
- Cited by: 26
- Wing, M. R., Devaney, J. M., Joffe, M. M., Xie, D., Feldman, H. I., Dominic, E. A., Guzman, N. J., Ramezani, A., Susztak, K., Herman, J. G., Cope, L., Harmon, B., Kwabi-Addo, B., Gordish-Dressman, H., Go, A. S., He, J., Lash, J. P., Kusek, J. W., & Raj, D. S. (2014). DNA methylation profile associated with rapid decline in kidney function: findings from the CRIC Study. Nephrology Dialysis Transplantation, 29(4), 864-872. https://doi.org/10.1093/ndt/gft537
- Cited by: 164
Leave a Reply