Can Under Methylation Cause Mitochondrial Dysfunction?
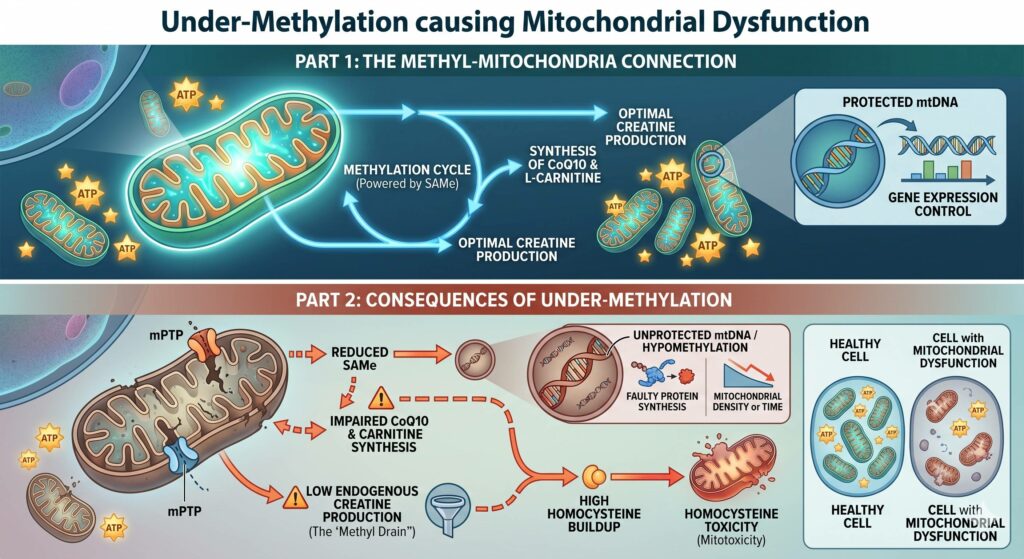
Yes, there is a profound biochemical link between under-methylation and mitochondrial dysfunction. Because the mitochondria rely on methyl groups for structural integrity, energy production, and DNA maintenance, a deficit in the methylation cycle (often involving low S-Adenosylmethionine, or SAMe) can significantly impair cellular respiration.
Here is how under-methylation contributes to mitochondrial issues:
1. Synthesis of CoQ10 and Carnitine
The mitochondria require two specific compounds to produce ATP (energy) effectively, both of which require methyl groups for their production:
- Coenzyme Q10 (CoQ10): This is a critical component of the electron transport chain. The final steps of CoQ10 synthesis require methyltransferase enzymes that use SAMe as a donor.
- L-Carnitine: This molecule “shuttles” fatty acids into the mitochondria to be burned for fuel (beta-oxidation). Carnitine synthesis requires the methylation of lysine, meaning under-methylation can lead to a “fuel delivery” problem.
2. The “Methyl Drain” of Creatine
Approximately 40% to 50% of your body’s methylation capacity is dedicated to producing creatine.
- Creatine is essential for recycling ATP in high-demand tissues like muscles and the brain.
- If the methylation cycle is sluggish (under-methylation), the body cannot produce enough endogenous creatine.
- This forces the mitochondria to work harder to maintain ATP levels, eventually leading to oxidative stress and “burnout” of the mitochondrial membrane.
3. Mitochondrial DNA (mtDNA) Protection
Mitochondria have their own DNA, which is much more susceptible to damage than nuclear DNA.
- Gene Expression: Methylation regulates the expression of mitochondrial genes. Under-methylation can lead to “hypomethylation” of mtDNA, causing the mitochondria to produce faulty proteins or fail to replicate properly.
- Repair Mechanisms: Without adequate methyl donors, the ability to repair oxidative damage to mtDNA is compromised, leading to a decline in mitochondrial density over time.
4. Homocysteine Toxicity
In under-methylation, homocysteine levels often rise because the body cannot efficiently convert it back into methionine.
- High levels of homocysteine are directly “mitotoxic.”
- Homocysteine can trigger the opening of the mitochondrial permeability transition pore (mPTP), which causes the mitochondria to swell and potentially rupture, leading to cell death.
Summary Table: Impact of Under-methylation
| Feature | Role in Mitochondria | Effect of Under-methylation |
| SAMe | Universal methyl donor | Reduced ATP production efficiency |
| CoQ10 | Electron transport | Increased oxidative stress (ROS) |
| Carnitine | Fatty acid transport | Reduced fat burning for energy |
| Creatine | ATP buffering | Rapid cellular fatigue |
| mtDNA | Genetic blueprint | Reduced mitochondrial biogenesis |
In short, when the methylation “engine” slows down, the mitochondrial “power plants” lose their ability to maintain their hardware and process their fuel, resulting in the systemic fatigue often associated with methylation imbalances.
Under-methylation significantly impairs mitochondrial function by restricting the availability of S-Adenosylmethionine (SAMe), the body’s primary methyl donor. This deficit directly hinders the synthesis of essential compounds like Coenzyme Q10 and L-Carnitine, which are necessary for transporting fatty acids and facilitating the electron transport chain. Furthermore, because nearly half of the body’s methylation capacity is dedicated to creating creatine, a sluggish cycle forces the mitochondria to work overtime to maintain ATP levels, eventually leading to structural “burnout” and systemic cellular fatigue.
Beyond energy production, under-methylation leaves the mitochondria vulnerable to genetic and chemical damage. A lack of methyl groups compromises the stability and repair of mitochondrial DNA (mtDNA), leading to poor replication and the production of faulty proteins. Simultaneously, the resulting buildup of homocysteine – a byproduct of impaired methylation – acts as a potent “mitotoxin” that can cause mitochondrial swelling and rupture. Together, these factors transform a metabolic imbalance into a state of chronic mitochondrial dysfunction, marked by increased oxidative stress and reduced cellular resilience.
Leave a Reply